China Is Now a Global Drug Innovation Powerhouse. Industrial Policy Had Little to Do with It.
China Is Now a Global Drug Innovation Powerhouse. Industrial Policy Had Little to Do with It. [ 5 min read ]
INSIGHTS
In 2010, China accounted for less than 8% of global clinical trials; by 2020, it had surpassed the United States in annual registered trial volume, reaching over 5,000 trials per year by 2024 — a 172% increase relative to the U.S.
Innovative trials targeting previously unexplored biological mechanisms rose by 49–123% relative to the U.S., and domestic Chinese firms account for 88% of the post-2015 increase.
The key driver was the 2016 National Reimbursement Drug List (NRDL) reform that traded drug price cuts for inclusion into China’s national insurance program, thereby massively expanding the market for innovative drugs, boosting cancer drug revenue by 500% and clinical trial activity by 86%.
The NRDL reform explains 43% of the surge in oncology trial activity — nearly double the combined contribution of knowledge accumulation and talent inflows from the U.S. (24%) — while industrial policies like Made in China 2025 played a negligible role.
Source Publication: Panle Jia Barwick, Hongyuan Xia, and Tianli Xia (2026). From Free Rider to Innovator: The Rise of China’s Drug Development. NBER working paper.
Read this brief on SUBSTACK
For most of the postwar era, the U.S. and Europe invented novel drugs, and the rest of the world consumed them. Developing economies had little incentive to invest in indigenous R&D — the fixed costs were too high, the markets too small, and the technological frontier too distant. China fit this model well into the 2000s, with its pharmaceutical sector focused on generic manufacturing and biosimilar production rather than frontier innovation. That changed abruptly in the mid-2010s as China rapidly moved up the value chain. What accounts for China's remarkable transition from pharmaceutical free rider to global innovator?
The data. The authors compare clinical trial activity and drug development outcomes between the U.S. and China before and after a healthcare reform expanded insurance coverage for innovative drugs. Trial data come from the Citeline TrialTrove database, a global census of clinical trial registrations; drug development outcomes I— including out-licensing deals and regulatory approvals — come from Citeline Pharmaprojects; and market size is measured using SinoHealth drug sales data at the province-quarter level. To rule out alternative explanations, the authors control for scientific knowledge accumulation (OpenAlex publications), international talent flows, regulatory streamlining (application records from China’s drug regulator), and industrial policy (State Council policy documents).
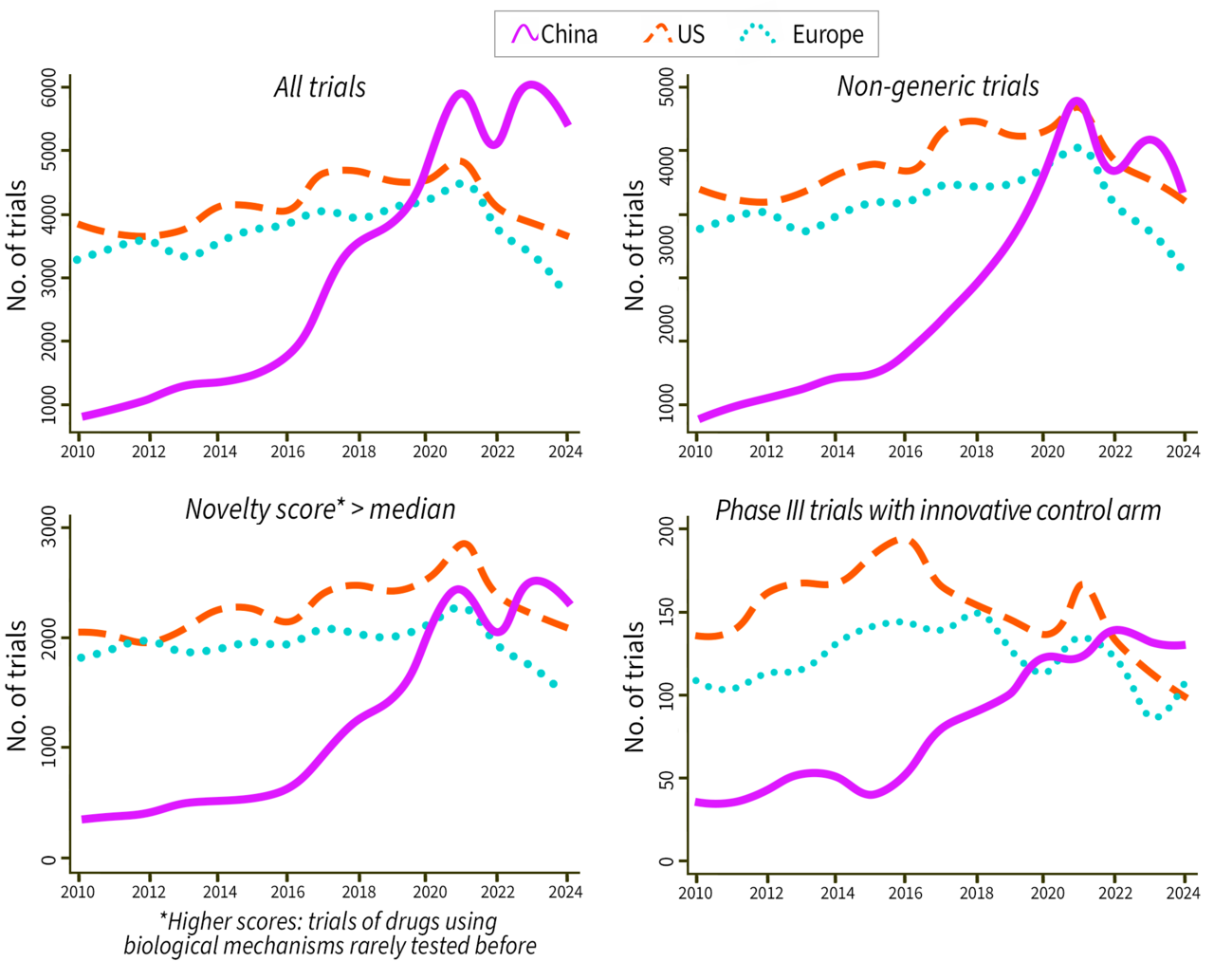
China's clinical trial surge was real and high quality. Before the 2016 National Reimbursement Drug List (NRDL) reform, China’s clinical trial activity tracked the U.S. but at a fraction of the scale, accounting for less than 8% of global trials in 2010. By 2024, that gap had not just closed but reversed: China's trial volume had grown by 172% relative to the U.S., surpassing it entirely by 2020. Importantly, this was not a surge in low-value generic testing. Three independent novelty measures — a mechanism-of- action-based novelty score, an AI-classified indicator of new molecular entities, and a measure of Phase III trials benchmarked against existing innovative drugs rather than placebos — all show increases of 49–123% relative to the U.S. Downstream outcomes tell the same story: out-licensing transactions to multinational corporations doubled, first-in-world regulatory approvals for Chinese-developed drugs rose 36%, and new firm entry grew 27%, all relative to 2015. Importantly, 88% of the post-2015 increase was driven by domestic Chinese firms, not multinationals relocating R&D to China — indicating genuine indigenous capacity building rather than geographic reallocation.
Clinical trial quantity and quality over time
The NRDL reform was the dominant driver. The authors identify the NRDL reform as the primary mechanism boosting China’s trials and drug development. Prior to 2016, China’s national insurance system covered mainly generic drugs, forcing patients to pay full out-of-pocket costs for innovative therapies. The NRDL reform introduced annual centralized price negotiations between the government and pharmaceutical firms, with successful inclusion triggering near-universal insurance coverage. While negotiated prices fell by an average of 50% (and 66% for oncology drugs), volumes surged by 350% on average and nearly 900% for cancer drugs. Net revenue for covered oncology drugs rose by 500%. The authors test whether disease areas that gained the most insurance coverage also saw the largest increases in R&D activity. Disease categories with greater coverage saw disproportionately larger increases in clinical trial activity, while a placebo test on disease categories never covered by the NRDL shows no systematic post-2015 increase.
Industrial policy played a marginal role. A careful decomposition exercise quantifies the relative contribution of five potential drivers. For oncology, the largest therapeutic area, the NRDL accounts for 43% of the observed post-2016 increase in clinical trials (roughly 439 of 1,004 additional trials in 2024 relative to 2016). The authors also tested whether the return of U.S.-trained Chinese scientists and growth in scientific publishing drove the surge — and both factors did show a positive effect. But together, talent inflows and knowledge accumulation explain only 24% of the post-2016 acceleration in trials.
Three other factors, including China's accession to international regulatory standards, a 2015 clinical trial application reform that cleared trial backlogs, and Made in China 2025 industrial subsidies collectively account for less than 1%. For Made in China 2025, the authors checked whether the trial surge was concentrated in cities that received targeted industrial subsidies. It wasn't — non-subsidized cities showed identical patterns of growth. This finding challenges the widespread assumption that China's technology ascent has been primarily driven by state industrial policy. At least in pharmaceuticals, the critical lever was expanding the market for innovative drugs, not funding their development with subsidies or R&D grants.
Good market design outperformed subsidies. In the short run, the reform generated approximately ¥22.5 billion per year in net benefit to society from expanded patient access to existing drugs. But the long-run gains — from entirely new drugs being developed — are conservatively estimated at three times that figure, roughly ¥66 billion per year. By credibly expanding the market for innovative drugs, the reform raised the expected return on R&D investment, pulling frontier clinical development into China. A well designed insurance reform that expands effective market size — particularly one that explicitly rewards clinical novelty in its coverage criteria — did more to catalyze frontier R&D than direct subsidies or grants.
